UNC Scientists on Trail of Perplexing COVID Variants
Posted on March 22, 2021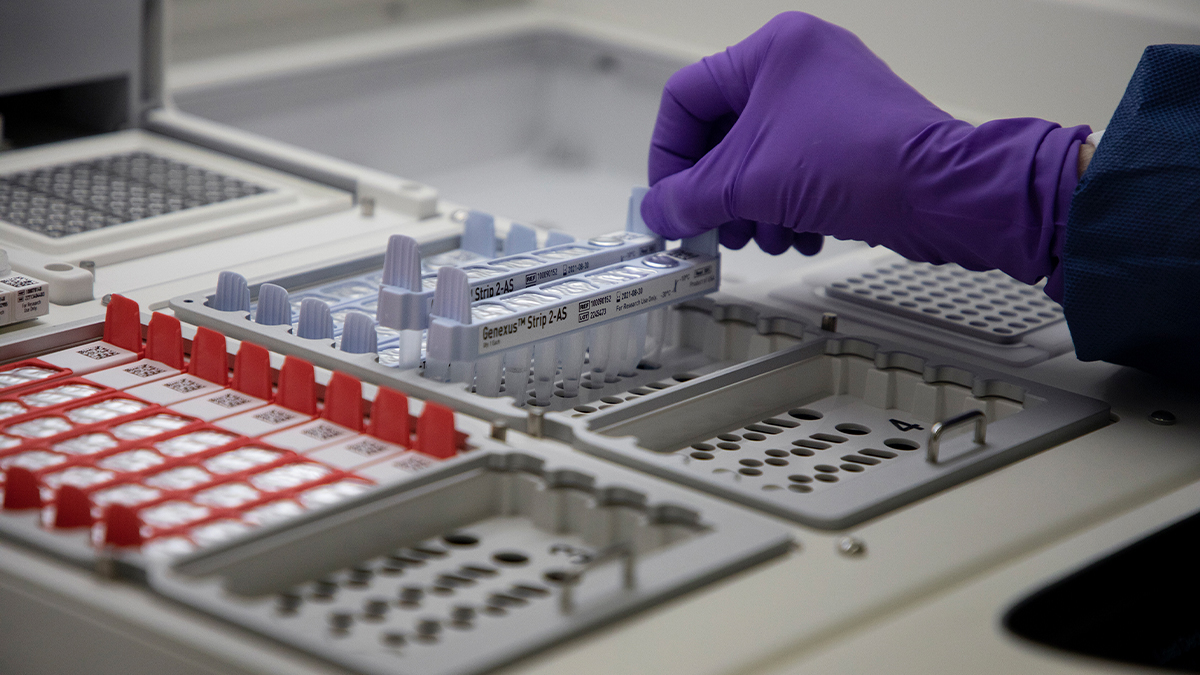
Linda Pluta prepares the Genexus Integrated Sequencer for genetic analysis of COVID-19 test samples. (UNC/Megan May)
Trays of tiny vials in hand, Linda Pluta stands in front of a shoulder-height machine, waiting for it to complete an ultraviolet light cleaning cycle. The timer beeps, and a plexiglass display case smoothly rises. She loads it with dozens of trays, presses a button, and the glass quickly descends. Soon the device comes to life, humming with enough force to make everything in the room buzz and vibrate.
The Genexus Integrated Sequencer may sound like something from a science-fiction movie, but it’s an everyday tool in the Dittmer Lab, where researchers are tracking variants of SARS-CoV-2.
Since fall, scientists have discovered variants from the U.K., South Africa and Brazil. Subsequently, all three have been reported in the U.S., and two have been found in North Carolina. Additionally, new variants have emerged in dozens of states throughout the U.S.
The issue isn’t that the virus is suddenly mutating — it’s that genetic sequencing has lagged far behind its spread. In the U.S., less than 1 percent of cumulative cases have been sequenced.
“That kind of makes you ask the question, ‘How many did we miss in between?’ ” says Fernando Rene Moreira, a postdoctoral researcher in the Gillings School of Global Public Health’s department of epidemiology.

UNC Researchers Dirk Dittmer, Melissa Miller and Ralph Baric.
As a virus replicates, mistakes are made in copying its genetic sequence. One theory suggests this is due to the frequency of virus replication; another points to factors in an individual host that cause these mistakes. It’s a classic example of evolution — if those changes are beneficial to the virus, they are more likely to persist as replication continues. The more SARS-CoV-2 spreads, and therefore replicates, the likelihood of these mutations increases.
“The number of infected people is tremendous, so there’s much more virus replication than ever before,” says Dirk Dittmer, a professor in the department of microbiology and immunology in the School of Medicine. “Every person that’s infected is a little test tube that allows the virus to change.”
While the terms “mutation” and “variant” may sound intimidating, it’s what viruses do. What concerns Dittmer and his colleagues is the number occurring at once.
“Normally when we sequence viruses, we find one or two mutations in the spike protein from the week before,” he said. “The U.K. one had nine new changes. That’s a huge jump and why everyone is worried.”
Carolina researchers Melissa Miller and Ralph Baric have joined Dittmer in a collaboration that acts as a SARS-CoV-2 surveillance pipeline. Miller is a professor of pathology and laboratory medicine and director of the clinical molecular and microbiology laboratories in the School of Medicine. Baric is a Kenan Professor in the Gillings School’s departments of epidemiology and microbiology and immunology.
Miller’s lab, which in mid-March was processing about 5,000 COVID-19 tests from UNC Health per week, sends positive samples to Dittmer’s, where they undergo genetic sequencing. Genomic anomalies are then passed to the Baric lab for further analysis in human cells and animal models. The goal is not only to detect new variants but also to understand where they first emerged, their pervasiveness and how they differ.

Fernando Rene Moreira, a postdoctoral researcher, works in the Baric Lab in the UNC Gillings School of Global Public Health. (UNC/Megan May)
Prior to the pandemic, Dittmer’s work focused on viral cancers. Twenty percent of cancers are actually due to a virus. For example, the human papillomavirus can cause cervical cancer, and the herpes virus can lead to the cancer Kaposi’s sarcoma. A big part of this research involves genetic sequencing.
“We built the infrastructure in the cancer center to hunt for new viruses,” Dittmer said. “So when COVID came, it was very easy for us to acquire samples from UNC Hospitals and start sequencing to identify which strains and variants are present in North Carolina.”
This endeavor started in May 2020. By that July, the lab got a hit. Dittmer’s team was able to trace the variant to virus mutations from someone returning to North Carolina from Seattle and another from a cruise to Italy.
Dubbed SARS-CoV-2 D614G, the variant was determined to be highly transmissible due to an increase of viral loads — the amount of virus — in the host and a mutation in the spike protein. This variant soon became dominant worldwide and is ancestral to the current list of variants.
Analysis in the Baric lab found the mutated spike protein — shaped like a “G” rather than the ancestral variant’s “D” shape —increases its ability to open and infect cells more efficiently. The good news is that studies in animal models have shown while the variant is more infectious, it does not cause worse symptoms, and its mutation may actually make it more vulnerable to vaccines.
As new variants are discovered, the threats they pose remain largely unclear. “I’ll let you know in two years,” Moreira joked. “Being trained in the sciences, you always have to wait for the data.”
Some current findings, though, do give insight into transmission and response to vaccines.
A recent study by researchers in the U.S. and U.K. reports the U.K. variant — B.1.1.7 — is 43 to 90 percent more transmissible than the ancestral strain. Data analysis from the lab testing company, Helix, found that it likely has accounted for more than 20 percent of new U.S. cases as of early March 2021. If the trajectory continues, some think the U.K. variant may be the dominant variant in the U.S. by the end of March.

The Genexus Integrated Sequencer processes COVID-19 test samples in the Dittmer Lab. (UNC/Megan May)
When it comes to vaccines, researchers believe that the U.K variant does not reduce the effectiveness of the Moderna or Pfizer shots. Data on the Novavax and AstraZeneca vaccines report efficacy rates of 85 percent and 74 percent, respectively.
Less is known about the variant originating in South Africa. Johnson & Johnson’s vaccine had a 57 percent efficacy in South Africa versus 72 percent in the U.S., and Novavax’s had 49 percent efficacy in South Africa versus 90 percent in the U.S. This discrepancy may be due to the low number of cases in South Africa compared to those in the U.S. However, testing in the Baric lab shows that the South African variant is about eight times more resistant to immunity elicited by current vaccines — supporting a critical need for continued surveillance.
Even with a reduction in efficacy, these vaccines all are proven to be very successful in fighting the virus and should not deter the public from vaccination, Dittmer says.
The COVID-19 relief bill includes $1.75 billion to better track and study SARS-CoV-2 variants. Researchers are working to develop clearer answers but need something not afforded to them during a pandemic — more time. While Miller’s lab processes thousands of COVID-19 tests per week, Dittmer’s can handle a maximum of 70. Once a sample containing mutations is passed to Baric’s lab, it can take months for analysis in human cells and animal models.
In the meantime, the best way to stop the emergence of new and potentially more dangerous variants is to stay vigilant with safety measures proven to stop the spread: wear a mask, stay physically distant and get vaccinated.
“It all depends on how good we are at continuing safety measures, which interrupt transmission,” Dittmer explained. “If you do that, then the pool of actively replicating viruses becomes so small that new variants simply don’t happen.”
This story was written by Megan May and was produced by Endeavors, UNC’s online research magazine. May is a photographer, videographer and writer for UNC Research and Endeavors websites.
